WHO prequalifies Bavarian Nordicĺs vaccine against Mpox
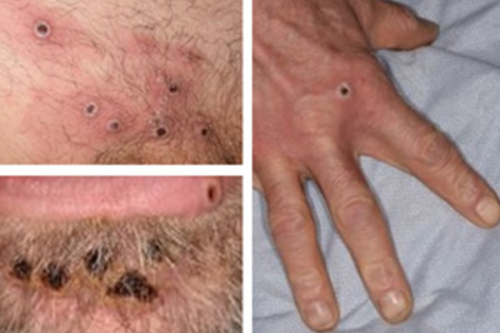
New Delhi, Sep 13 (IANS) Amid the global outbreak of Mpox, the World Health Organization (WHO) on Thursday announced prequalifying Bavarian Nordic’s vaccine -- a first against the deadly monkeypox virus (MPXV).
Modified Vaccinia Ankara-Bavarian Nordic or MVA-BN is indicated for active immunisation against smallpox, Mpox, and related orthopoxvirus infections and disease in all adults 18 years of age and older.
The vaccine can be administered as a 2-dose injection given 4 weeks apart.
“Available data shows that a single-dose MVA-BN vaccine given before exposure has an estimated 76 per cent effectiveness in protecting people against Mpox, with the 2-dose schedule achieving an estimated 82 per cent effectiveness,” the WHO said in a statement.
“This first prequalification of a vaccine against Mpox is an important step in our fight against the disease, both in the context of the current outbreaks in Africa and in the future,” said WHO Director-General Dr Tedros Adhanom Ghebreyesus.
Ghebreyesus stressed the need to "scale up in procurement, donations, and rollout to ensure equitable access to vaccines".
In addition, other public health tools are also urgently needed "to prevent infections, stop transmission, and save lives”, he added.
The WHO prequalification comes even as the UN health body last month declared a global health emergency over its outbreak in Africa. The prequalification may facilitate timely and increased access, to contain transmission and help contain the outbreak.
The WHO’s assessment for prequalification is based on information submitted by Bavarian Nordic, and reviewed by the European Medicines Agency, the regulatory agency of record for this vaccine.
The MVA-BN is currently not licensed for persons under 18 years of age, but the WHO recommended its use “off-label” in infants, children, adolescents, and pregnant and people with weaker immune system in cases where the benefits of vaccination outweigh the potential risks.
MVA-BN is the only non-replicating Mpox vaccine approved in the US, Switzerland, and Singapore (marketed as JYNNEOS), Canada (marketed as IMVAMUNE), and the EU/EAA and UK (marketed as IMVANEX).
Meanwhile, more than 120 countries have confirmed over 103,000 cases of Mpox since the onset of the global outbreak in 2022.
In 2024 alone, there were 25,237 suspected and confirmed cases and 723 deaths from different outbreaks in 14 countries of the African Region.

|

|

